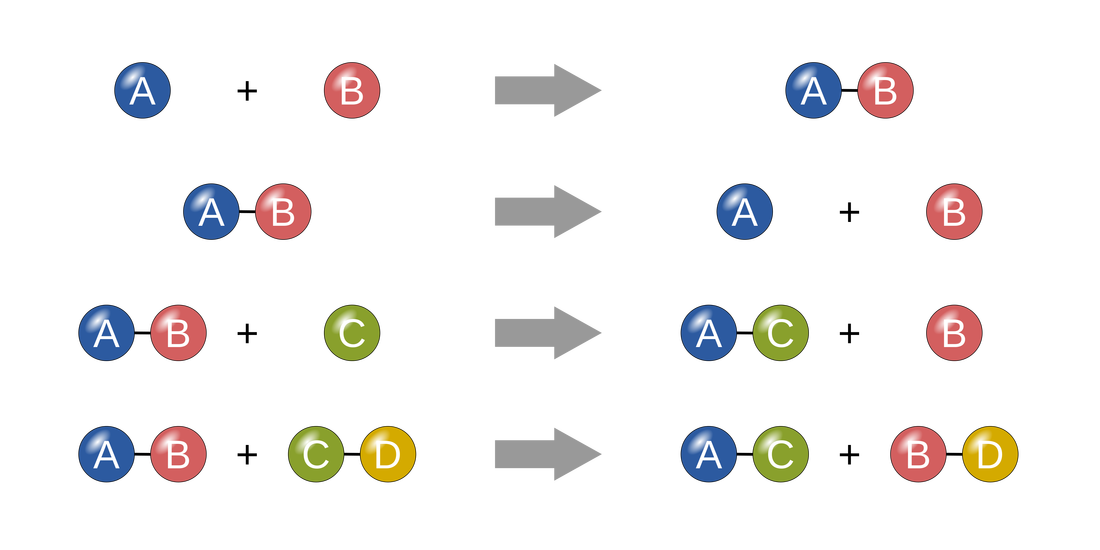
Chemistry Vocabulary Words
 Acid: Proton Donor
Acid: Proton Donor
Acid:
- A substance which neutralizes bases (alkalis);
- Can be corrosive, able to dissolve metals;
- Sour-tasting (lemons and other citrus fruit contain ascerbic acid);
- pH of less than 7
- In chemical reactions, an acid can donate a proton (H+) or accept an electron pair.
|
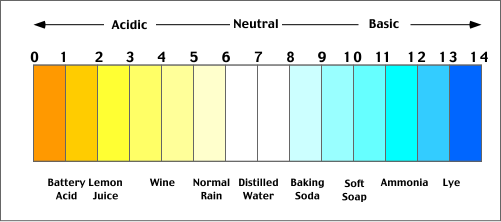
pH
pH is a measure of hydrogen ion concentration; a measure of the acidity or alkalinity of a solution. Aqueous solutions at 25°C with a pH less than seven are acidic, while those with a pH greater than seven are basic or alkaline. A pH level of is 7.0 at 25°C is defined as 'neutral' because the concentration of H3O+ equals the concentration of OH− in pure water. Simple: a scale that measures the acidity of a solution. |